Liaoning Haitai Scientific and Technological Development Co.,Ltd
Marketing office:31-floor 1 unit building No.1 Poly Ray New City, Wanghua District Fushun, Liaoning Province, China. Fctory: No.38 Qingtaizi Road, Shenfu New District, Liaoning Province, China
Post Code:113122 E-mail:sales@chinacatalyst.com
辽ICP备2022002379号-1 Powered by 300.cn ShenYang Mobile E-mail
TENDENCY OF ISOMERIZATION PROCESS DEVELOPMENT (3)
IS-614A catalyst is one of the first developments of Axens. ATIS-2L catalyst,which is the product of teamwork with Akzo Nobel was developed later on its base.ATIS-2L distinguishes oneself by higher activity (octane number of the isomerizate is one point higher), lower apparent density (catalyst charge decreases by 22 %), lower by 10 % platinum content [8, 12]. So, its application is economically more attractive solution. The first industrial loading was in 2003 year [9, 13].
Main licensors of this process in Russia are Co Ltd Scientific production firm“Olkat” (НИП-3А), JSC “VNIINeftehim” (ИП-05) [5].Sulfated metal oxides based catalysts get heightened interest last years as they
combine main advantages of medium-temperature and low-temperature catalysts. They are active, resistant to poisonous impurities and able for regeneration. The only drawback, as for zeolite catalysts, is necessity in compressor for recycling of hydrogen-rich gas (Figure 5).
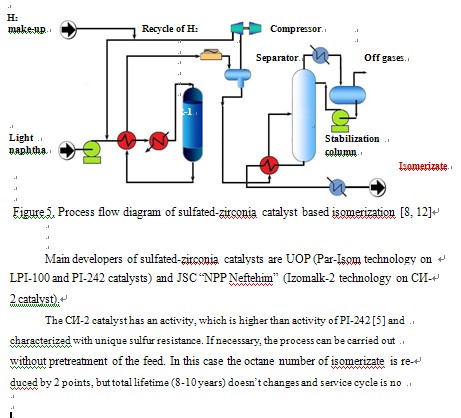
less than 12 months. The feed may contain considerable quantity of benzene which is
hydrogenated efficiently on the catalyst. The CИ-2 catalyst is produced in JSC
“Industrial catalysts” (Ryazan) and JSC “Angarsky factory of catalysts and organic
synthesis” by license of JSC “NPP Neftehim” [8].
The Pt/WO3-ZrO2 catalyst, which is developed in the University of Hokaydo city
(Japan), shows higher activity and selectivity in isomerization reaction of n-alkanes
compared to sulfated-zirconia catalysts. The advantage of this type of catalyst is ex-
plained by rapid surface diffusion of hydrogen atoms, which are converted into protons
and hydrides on the Lewes acid sites, thereby increasing catalyst activity and selectivity
[14].
5. TECHNOLOGIES SCHEMES OF ISOMERIZATION PROCESS
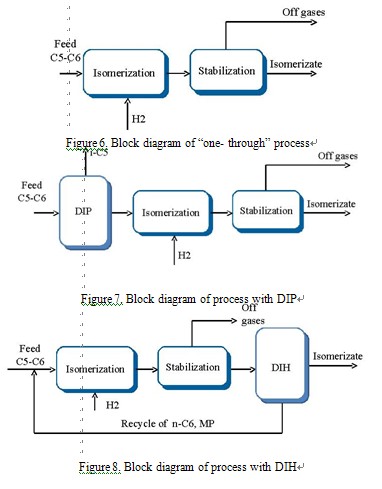
Economically efficient “one- through” scheme without any recycle can be used with minimum investment in realization of isomerization process (Figure 6). The scheme with deisopentanizer (DIP) before the reactor section allows the producing of isomerizate with high octane number, increasing of conversion level of npentanes and reducing the reactor duty simultaneously. The technology is reasonable in the case of isopentanes content in the feed more than 13-15 % (Figure 7) [9,13,15].
The scheme with deisohexanizer (DIH) after the isomerization reactor is the simplest way to produce the isomerizate with higher octane number. In this case nonconverted low-octane components (methylcyclopentane and nhexane) are recycled into reactor. However the given scheme allows only increasing of hexanes conversion, but doesn’t raise the content of isopentanes in the product (Figure 8). The scheme of the process may include both deisopentanizer and deisohexanizer (with DIP and DIH)[9,13,15].
Scheme with recycle of n-pentane (with DIP and DP) requires providing with depentanizer of isomerizate after the reaction section and deisopentanizer before the reactor. Schemes with recycle of n-pentane and n-hexane. For total conversion of all linear paraffins (not only n-С6 but also n-С5) into isomers, their total recycle is necessary, which can be realized by set of distillation columns (with DIP, DIH and DP) or by adsorption on molecular sieves.
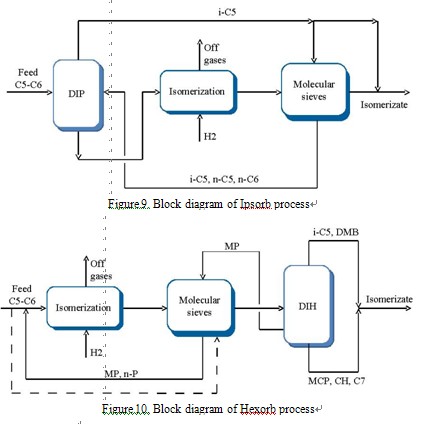
8The method of adsorption on molecular sieves (in liquid or vapor phase) is based on capability of pores with definite size to adsorb selectively the molecules of n-paraffins. The next stage is desorption of n-paraffins from pores and its recycle to the feed stock.
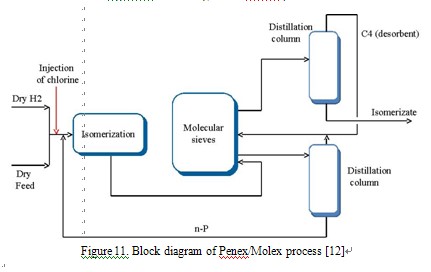
Stages of adsorption and desorption are repeated in cycles or pseudo-continuously. Axens offers two patented isomerization processes on the molecular sieves:Ipsorb and Hexorb (Figure 9, 10). UOP offers the processes with adsorption systems on the molecular sieves in vapour phase (Penex/Iso Siv) and liquid phase (Pe-nex/Molex (Figure 11)), and process, which combines adsorptive separation of unconverted nparaffins from isomers and deisohexanizing Penex/DIH/PSA [5, 16].